Evidence of ongoing genetic engineering Large-scale experiments with the unsuspecting
Tue 1:35 pm +00:00, 1 Nov 2022
Some sources for the work I mentioned about non-random toxicity fluctuations of the so-called “Vaccines” from Pfizer, Moderna and Janssen can be found here at Craig Paardekooper:
https://www.howbadismybatch.com/
January 9, 2022 (changes and additions last March 11, 2022)
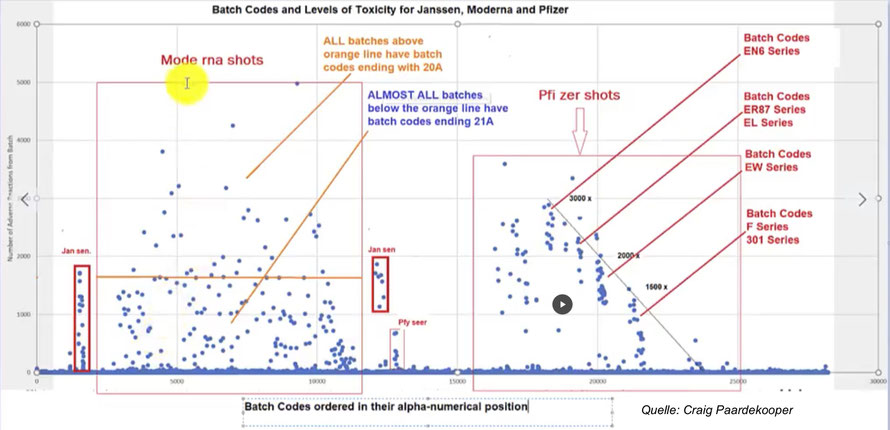
Craig Paardekooper and others have conducted a detailed analysis of the US database VAERS, in which the damage is documented in close temporal relation to the administration of the preparations from BioNTech/Pfizer, Moderna and Janssen. It turned out that the individual batches of the so-called vaccinations have extremely different toxicity levels. Some batches have increased toxicity up to 3000 times. The decision to vaccinate becomes Russian roulette.
The deviations are so extreme that they cannot be due to random or application-related toxicity fluctuations. Rather, there is some evidence that large-scale genetic engineering tests are currently being carried out on the broad, unsuspecting population under the protection of the alleged emergency and that this has been made possible, even promoted, by the legal-political preparatory work and the help of the governments and authorities.
The tried and tested rules, which required careful, long-term studies for the approval of a drug, were suspended under the pretext of a pandemic. Many things can now be tried out and plenty of use is made of them. Pharmaceutical companies are currently using this unprecedented opportunity to test more than 120 experimental corona vaccine candidates in an unbureaucratic manner. Bayer boss Stefan Oelrich (video after 1:37:40 h) said in his speech at the World Health Summit 2021 in Berlin described the mRNA vaccine as “gene therapy that 95% of the population would have rejected two years earlier”. Riding the wave of fear, profit-oriented pharmaceutical companies are currently trying out every method and product that can be patented and, thanks to the parallel data collection campaigns, it is very easy to observe the effects of their experiments – without having to be held liable for them. Resistance by ethics committees excluded.
The gateway for the experimental pharmaceutical industry is the so-called “telescopic approval process”. If the development of new vaccines otherwise took many years (specifically at least five years, an average of eight years) and proceeded according to strict graduated rules, the WHO made the “telescopic approval procedure” possible by declaring a “pandemic emergency”.
According to the current practice of clinical studies, there were at least four phases, each of which had to provide the required safety level for the next stage, compare the statements on the website of the Federal Ministry of Education and Research :
- Phase I trials are small studies in which a new treatment is used in humans for the first time, in healthy volunteers. At this stage, basic properties such as tolerability and safety of a new drug are checked to see if it is suitable for use in humans.
- Phase II studies are slightly larger than Phase I studies. They usually have 100 to 300 participants. In phase II, a drug is tested for the first time in patients suffering from the disease for which the drug is being developed. It’s all about the optimal dosage. In addition, initial data on effectiveness are collected.
- Phase III trials are large trials. They provide relatively precise information about effectiveness and tolerability. In the vast majority of cases, these are comparative studies. Patients receiving the treatment under investigation are compared with a control group receiving a different treatment.
- Phase IV trials take place when a drug is already on the market. There are different reasons for phase IV studies. For example, it can make sense to re-examine a drug that has already been approved in patients with certain properties. In phase IV studies, rare side effects of a drug can also be better assessed because more patients are being treated.
Actually, we should be in a phase IV study (post-marketing/observation study) for the syringes from Moderna, BioNtech-Pfizer, Janssen or AstraZeneka after their “conditional market approval”. The competent drug authority in Germany, the Paul-Ehrlich-Institut (PEI) explains the conditional approval – in italics: author’s comments ):
“A conditional approval is an approval that is linked to conditions. It can be granted in the public interest for a medicinal product,
- when the benefit of immediate drug availability outweighs the risk of less extensive data than would normally be required. (Where is the benefit-harm analysis?)
- – when it comes to the treatment or prevention of a life-threatening disease. This includes medicines for rare diseases ( COVID-19 has not caused more illnesses and deaths than normal flu )
- if the CHMP determines that all of the following requirements are met:
- A positive benefit-risk balance of the product, ie the benefit to public health from the immediate availability of the drug on the market, outweighs the risks that exist due to the planned subsequent submission of further data. ( is not verifiable and has not been verified )
- The applicant will provide full data at a later date. ( what? when? see VERS data) )
- An unmet medical need is met ( this is obviously not the case, in fact it has been and is massively mistreated and this causes damage in the first place ). Conditional approvals are valid for one year and can be renewed annually. You can switch to full approval.
Although the study phases are pushed together in the “telescoped” procedure, it must of course already be determined which components are to be contained in a candidate to be tested, and all approved drugs must have a correspondingly identical content. Reserve samples of each batch should document this as well as regular controls by the drug authorities.
However, when asked, the PEI stated that it does not carry out these drug control tests itself, but relies on the prescribed quality controls and reports that the manufacturers are obliged to provide.
To date, the PEI has not responded to a request from October 15, 2021 regarding the Freedom of Information Act regarding the content monitoring of corona vaccine batches. As with other corona measures, evidence and transparency are apparently not required.
In the meantime, several international research teams have systematically examined the USA side effect database VAERS and found as early as October 31, 2021 that all serious side effects and deaths reported in the USA only relate to a very small part of the batches (batches or lots) ( Here is a report from the official VAERS website ). More and more such results are now becoming known and reveal frightening connections.
The VAERS database provided evidence of batches of vaccines with very different effectsG. It includes a record of the reported adverse reactions associated with each batch. So an obvious task was to create a graph showing how the toxicity of batches varied over time and location throughout 2021. Diagrams do not show with certainty when the toxic batches were used, but how toxic they were. There are also indications that the participating pharmaceutical companies apparently acted in concert. (In order not to interfere with each other’s given time window?) Finally, one can even guess at the purpose of these distributions, e.g. B. examining the effects of different doses (type of harm and deaths), etc.”
Former Pfizer Research Director Mike Yeadon says:
“As for intent to cause harm, including death, I’m convinced. I’m not alone either: several completely independent analysts agree on these points:
1. Several of us feel that the whole “hot lots” situation indicates intent, but the data needs to be well understood. Craig Paardekooper’s original analysis is flawed in one important respect. He mistakenly, but understandably, equated ticket numbering with chronological order. That is not correct. So these patterns, which look to me like a dose-response relationship exemplified over time, with companies appearing to coordinate to avoid each other, are arising as a result of this unsubstantiated assumption.
2. However, these lot numbers and associated serious adverse reaction values are REAL and they exist in VAERS. For example, Pfizer can pull the data for its lots and plot it against the SAE (severe side effect) rates, and it would produce graphs very similar to the Paardekooper analysis.
3. We believe this is intentional because the patterns of SAEs associated with lot numbers are not random. The variability of the SAEs per lot is gigantic and cannot be explained by harmless factors. For example, product instability and deterioration cannot produce these effects. In general, degradation results in a loss of activity rather than the acquisition of increased toxicity. One could argue that this is perhaps the exception to the rule. I show that this is not possible because the same extraordinary pattern, that a small percentage of batches are extremely toxic, is observed for three products with two technologies (mRNA and DNA). No: This is intentional and must be known to the company.
4. The adverse events per batch are orders of magnitude greater than for any comparable product (flu vaccine) and the variability from batch to batch is such that it cannot be assumed that all vials contain the same product. 5. We have checked the batch sizes for 33 Pfizer batches and there is no or very little correlation with batch size – clearly something different here. ( emphasis WW )
6. It follows that these products should be considered FALSE, whether intentional or accidental. In particular, Pfizer cannot demonstrate that what they advertise as their product is actually in the vial and consistent between vials. This would already be a crime, even if there was no intent.” ( personal communication )
What is alarming is that all three companies are conducting similar studies with greatly increased toxicity . Apparently they do it in such a way that they don’t get in each other’s way and apparently distribute and “dilute” their toxic experiments in such a way that it is hardly noticeable at first glance.
The dose-finding studies now suspected in the USA should usually have been completed long before the phase III approval studies (see above). They should be undertaken as phase II studies with a very limited number of well-informed volunteers.
Hence my urgent suspicion that the wrong pandemic is being used to try out a lot of things that would otherwise be far too risky and not allowed.
The intermediate “saline batches” would have five effects for the companies:
1. They dilute the otherwise alarming side effects
2. they cost little and still bring full price,
3. They provide the control groups that Big Pharma otherwise had to pay dearly for in Stage 2 or 3,
4. They are also fully financed from taxpayers’ money and
5. The risks of these “telescopic” studies are borne by the public sector.
However, we now have clear indications from VAERS that study structures in the state-prescribed and financed mass vaccinations with completely new products from Biontech, Janssen and Moderna will only be carried out extensively and planned after approval.
This is forbidden and punishable by law and clearly violates the Nuremberg Code and all relevant laws for conducting studies or marketing drugs. It is obviously not an oversight or neglect of quality but a planned offense. In this phase of institutional corruption, many companies sense huge opportunities and are already announcing to their investors that the high-risk health mRNA hype will continueon. Naturally, the primary interest of a drug company is economic success and not health benefits. Whether other substances such as graphene oxide or other nanoparticles also play a role in the ongoing telescoped studies has been discussed by many for a few months after they were found in the Covid-19 syringes. Many people have lost confidence in the reliability of study results or even drugs that are already on the market. That seems justified to me, because even the search for counterfeit or counterfeited (counterfeit or adulterated) medicines by an Interpol department specially set up for this purpose is co- financed by the pharmaceutical industry.
Under the term ” telescopic procedure” , the security levels of the study phases with official approval are undermined in “Corona”. But not only that.
Also the strict monitoring that is otherwise usual in phase IV (post-marketing).and transparent, scheduled documentation of batch-related content controls is apparently left entirely to the sponsors, i.e. the pharmaceutical companies. They are even allowed to use new formulations (nucleic acid sequences?) under the pretext of mutating pathogens. In a non-transparent procedure, everything can apparently be tried out on everyone without an ethics committee or even the affected patients being able/must agree informed about the risks or the state of research and its risks. A corresponding explanation of the millions of subjects does not take place in any case. They are even forced to participate without being informed. All of this was never allowed and constitutes a crime such as that which was the subject of the Nuremberg trials.
The invention of the telescoping method is presented as a trick at the expense of safety. However, this trick becomes a crime when millions of unsuspecting people have to risk their lives in the process.
Craig Paardekooper, one of the researchers, has put a database online, but it’s difficult to find on Google. At https://www.howbadismybatch.com/ you can now check for yourself which batches led to how many side effects or deaths.
For self-protection, anyone who wants to undergo the so-called corona vaccination despite the now known high potential for damage should ask their doctor or pharmacist before the injection what documented effect the batches they use have. If doctors and pharmacists have to investigate at this point, there is a chance that they will become aware of their responsibilities as potential liability.
Addendum: The importance of a batch expiration date
So we
have a list of expiration dates for all vaccine batches . What the hell! Well, here’s why this list is important…
The government provides physicians with a list of expiration dates for all batches of vaccines. However, the CDC keeps this list secret from the general public (why?). A contact passed this list on to me. It has struck me that the lots on the expiration list are ALL those with the highest number of adverse reaction (ADR) reports in each alphabetical group.I wondered why is that? Why weren’t any of the other games on the expiry list – the ones with only a handful of reports? (I had previously suspected that the others might be placebos.)
Then it occurred to me that placebos don’t expire. Salt water does not expire. So it has no expiration date. Only the biologically active batches will be on the expiration list. That’s possibly why the CDC didn’t want the public to see this list. They only give it out to medical professionals – for “safety” reasons – because it shows which are placebos and which are biologically active.
If a batch isn’t on the list, is it a placebo? (question mark from Wodarg)
So now we have a list of all biologically active batches. This is of course good information for people who want to avoid toxic batches. Just check your batch against the expiration list. If it is not listed there, it is most likely a placebo.
Note: Please keep in mind that new batches are introduced which may not be on the existing list and that even among biologically active batches there are still significant differences in toxicity – one bad batch may not be as toxic as another .
Note: This expiration list may only apply to US lots. It may not apply to European batches, which often have different batch codes.
Remark Wodarg:
And again the request, if necessary, talk to your vaccinator about these batches
and if necessary, insist on a batch with few side effects.